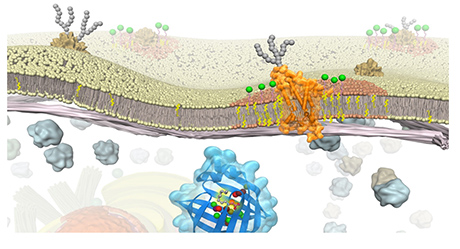
The main components of cell membranes are lipids, proteins, and sugar chains. Turning chemistry’s eye to complex self-aggregates
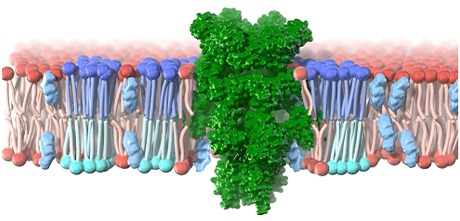
Biological membranes are extremely complex molecular complexes consisting of lipids, proteins, and sugar chains. The lipid bilayer, the main body of the membrane, is a self-assembly of small molecules such as phospholipids, glycolipids and sterols. Compared to membrane proteins, the roles of lipids and carbohydrates in biological membranes remain largely unknown. In this project, we will conduct cross-disciplinary research to deepen our understanding of biological phenomena based on the elucidation of functions in cell membranes by promoting the integration of chemistry and life sciences. In terms of application, we aim to return the research results to society by collaborating with researchers outside the university in the biomedical field in analyzing the composition of cell surface layers.
FEATURE
This is an interdisciplinary research project in which teams specializing in synthetic organic chemistry, glycoscience, and instrumental analysis collaborate with each other.
We will clarify the functional structures of soft biomolecules such as lipids and sugar chains, which have not been elucidated so far.
We will elucidate precisely how biomolecules interact with proteins to bring out their biological functions.
This approach has the potential to be a stepping stone to new drug discovery.
RESULTS
Research progress
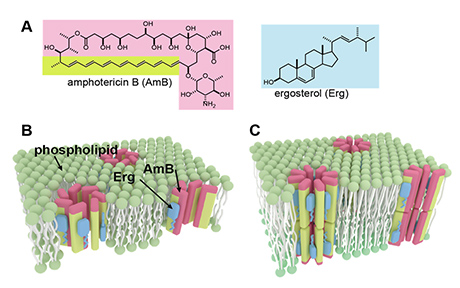
Elucidation of lipid recognition mechanism by chemical approach
Amphotericin B, an antifungal drug used for many years, forms fungicidal ion-permeable channels on the cell membrane. We have experimentally clarified the three-dimensional structure of the molecular assembly that this drug forms on hydrated membranes in the presence of the fungal sterol ergosterol (A right). As a result, seven drug molecules stably assemble to form an ion-conducting channel like B, unlike the previously considered C. Based on the obtained structure, we proposed a mechanism in which the formation of a complex with ergosterol stabilizes the drug aggregates, causing channel aggregation and enhancing pharmacological action. The elucidation of the channel structure allows us to understand that the drug is selectively toxic to fungi rather than human cells.
Further development
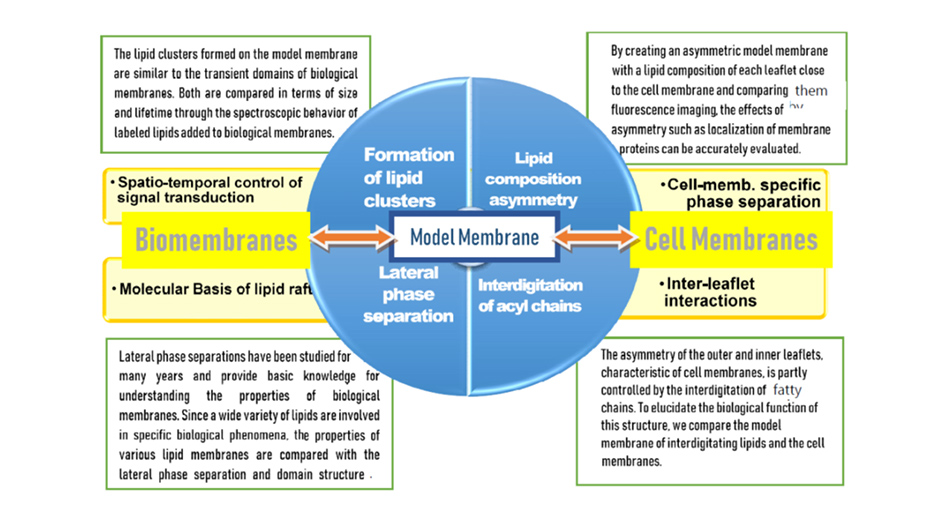
Structure-Function Relationships of Lipid Molecules in the Quadruple Layer of Biological Membranes Including Hydrated Layer
Research on biomembranes has lagged behind that on membrane proteins, which has made remarkable progress in recent years, and the formation mechanism and role of domain structures (lipid rafts) caused by the localization of membrane lipids are not well understood. In order to solve this problem, it is desirable to create model membranes that functionally reproduce biological membranes. However, homogeneous artificial membranes of simple composition have not been able to reproduce the domain structure unique to biological membranes, which is localized and short-lived. In order to solve this problem, it is necessary to accurately understand the interactions between the outer and inner leaflets of biomembranes, which have different lipid compositions, and between them and the hydration layer (a total of four layers, as shown in the figure on the right). In this project, we will reproduce the asymmetry of the outer and inner leaflets, the localization of phospholipids and glycolipids, and the molecular interactions at their interfaces, and compare them with those of biological membranes. By extracting the unique features of biomembranes and elucidating the molecular picture of lipids, we aim to understand biomembranes essentially.